Washington, DC— Every school child learns about the water cycle—evaporation, condensation, precipitation, and collection. But what if there were a deep Earth component of this process happening on geologic timescales that makes our planet ideal for sustaining life as we know it?
New work published in the Proceedings of the National Academy of Sciences by Carnegie’s Yanhao Lin and Michael Walter—along with former Carnegie scientists and ongoing collaborators Ho-Kwang “Dave” Mao and Qingyang Hu of the Center for High Pressure Science and Technology Advanced Research Shanghai and Yue Meng of Argonne National Laboratory—demonstrates that a key mineral called stishovite is capable of storing and transporting large amounts of water even under extreme conditions like those found in Earth’s lower mantle.
This is important, because it shows that substantial quantities of water could be present farther into the mantle than previously thought, indicating that a whole-mantle water cycle is possible.
“To get down into the mantle, water must be incorporated into minerals on the surface and then be stably maintained in those structures under the conditions found deep inside the planet,” explained lead author Lin.
The researchers used lab-based mimicry to study the mineral stishovite, which is a high-pressure form of quartz, when it’s with water under high pressure and temperature conditions. We already know substantial amounts of water can be stored in silicate minerals in the Earth’s upper mantle, which exists between 100 and 670 kilometers (or 62 to 416 miles) deep. But the team examined stishovite and water under simulated conditions like those found deep in the lower mantle, which exists between 670 and 2,900 kilometers (or 416 to 1,802 miles) down, where it was thought that much less water could be stably stored in minerals.
“Stishovite is a silica-based mineral and a major component of the oceanic crust.” explained Mao. “In plate tectonics, there are areas called subduction zones where an oceanic plate slides beneath a continental plate, sinking from the Earth’s surface into its depths. When this happens, stishovite is transported into the mantle.”
The discovery of diamonds with water-containing mineral inclusions that come from depths to about 700 kilometers (or 535 miles) down in the mantle revealed that water indeed gets at least that far down if it finds the right mineral with which to hitch a ride.
Stishovite is one such mineral, but is it capable of taking water even deeper, down into the lower mantle? This is what the researchers set out to discover.
They subjected tiny samples of stishovite with water to a range of about 320,000 to 510,000 times normal atmospheric pressure and heated it to a range of about 1,000 to 1,500 degrees Celsius simulating a gradient transiting from upper mantle conditions to lower mantle conditions. Remarkably, they found that stishovite can accommodate large amounts water even under these conditions.
“If water can be stored in minerals at lower mantle pressures and temperatures, it could indicate that there is a global water cycle occurring on very long geologic time scales,” explained Walter. “This could alter our understanding of how deep planetary interiors may influence or control the water content at the surface.”
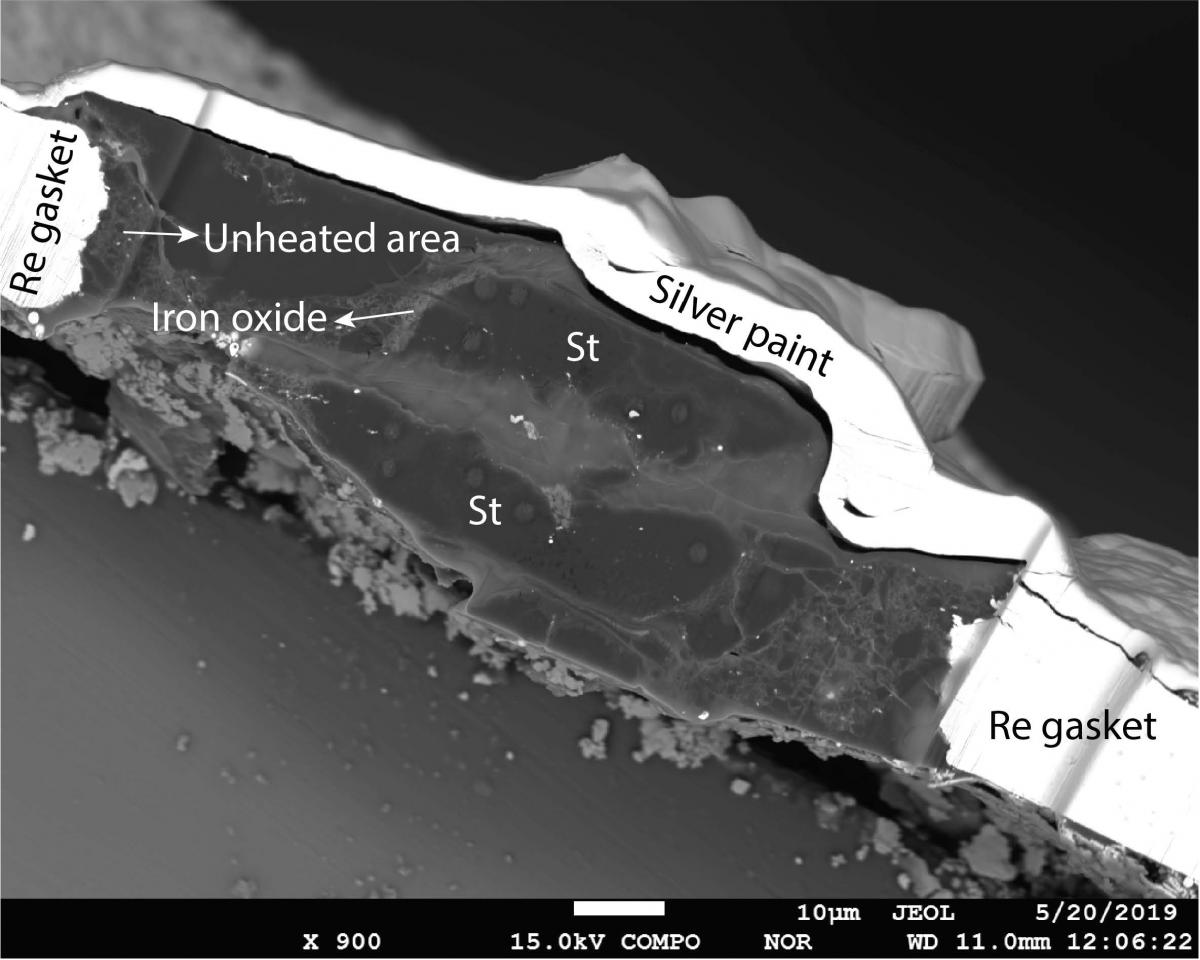
Image Caption: A tiny sample of stishovite used by the researchers in the lab. Photo is provided by Yanhao Lin.
__________________
This work was supported, in part, by the U.S. National Science Foundation.
